Member-only story
At-Home Coronavirus Testing Could Be Coming Soon
Tech startups want to provide self-swab kits, but the FDA has stepped in
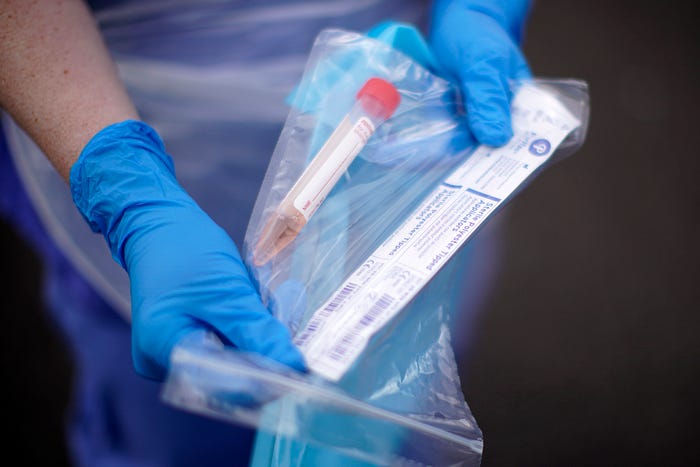
 You may soon be able to swab your nose at home and send the sample away to get tested for coronavirus without having to leave your couch — if U.S. government officials allow it.
You may soon be able to swab your nose at home and send the sample away to get tested for coronavirus without having to leave your couch — if U.S. government officials allow it.
Last week, a handful of telemedicine companies announced plans to make at-home collection kits available to patients suspected of having Covid-19. Tech startups like Nurx, Carbon Health, and Everlywell were set to ship thousands of at-home testing kits to customers this week.
But both Nurx and Carbon Health paused sales of their tests after the Food and Drug Administration, which oversees diagnostic tests, issued a strongly worded statement on March 20 warning consumers against what it called “unauthorized fraudulent test kits.” The agency didn’t specify which companies were the target of the notice. Everlywell decided to make its tests available only to hospitals and health care providers for now.
“At this time, the FDA has not authorized any test that is available to purchase for testing yourself at home for COVID-19,” FDA officials said in the statement. They noted that these tests can pose health risks because they could keep some people from seeking proper medical care.
With coronavirus tests still in short supply around the United States, at-home testing could help people with mild symptoms learn if they have Covid-19 without having to leave home and risk infecting others. They could drop their sample in the mail and receive a result in a few days without even having to interact with a health care worker. Health officials have stressed that widespread testing is crucial for tracking and controlling the pandemic.
That’s part of the reason why, in February, the FDA allowed commercial labs to start developing their own coronavirus tests when it was clear that more testing would be needed around the country.
Nurx, Carbon Health, and Everlywell took that as a green light to make their own tests and get them out to people who health care providers believe have Covid-19 but have been instructed to stay home. Around the country, testing is recommended for at-risk groups like the elderly and those…

